 The first two groups (1A and 2A) along with helium are called the s block. The periodic table is organized into blocks to represent the orbitals. It's confusing, but technically it has more energy, so it comes after On a side note, yes, I know 3d orbitals comes after 4s. If you add up all the numbers after the orbital letters, you'll find they equal twenty-six. That is the electron configuration for iron. Iron has an atomic number of 26, so that mean's it'll have 26 electrons in its neutral form. Hopefully you're following me, but the best way to really get the hang of this is to practice it yourself. So oxygen's electron configuration is: 1s2 2s2 2p4 so then we would say 2p4Ĥ = the number of electrons needed to complete oxygen's number of 8 :max_bytes(150000):strip_icc()/GettyImages-604346724-589a56263df78caebc80b0c5.jpg)
Do you remember me saying that up to three p orbitals can exist in each energy level? Well that means that there can be up to six electrons in each energy level for the p orbitals. However, we still only have half the electrons. The s orbital there is also filled completely for 2s2. Now, there can be no p orbitals in the first energy level, so then we jump straight to the second. So on the first energy level, we have 1s2, because it has at least as many electrons as helium. Okay, so we know oxygen has 8 electrons, and only two electrons can exist in each orbital. This means thatĪnd if you were to go a step farther with helium, you would get 1s2, because it has two electrons in that s orbital. Hydrogen has an electron configuration of 1s1. Their electron configuration is determined by the number of electrons their neutral form has and the orbitals in which those electrons exist. The numbers that you are talking about are called electron configurations, and each element has its own unique one. And I really don't know much about f orbitals (I've never had one that large) but I believe there can be seven d orbitals are a really complicated shape and there can be up to five. p orbitals are kind of dumbbell shaped and there can be up to three of them at each energy level. s orbitals are sphere shaped, and there can only be one at each energy level. Okay, now, there are different types of orbitals that each take different shapes. Finally, each orbital can hold only two electrons and no more. The farther out the orbital from the nucleus, the more energy the electron has. Each orbital also represents a different energy level. For example, if the orbital is in the shape of a sphere, the electron can exist in any point within that sphere, but it cannot go outside that sphere. An orbital is basically a given space that an electron can exist in. You may already know some of this stuff but I'll go over all of it anyway.īasically, every atom has orbitals around their nucleus. These noble gases are highly stable and do not react to form compounds under normal conditions.ģ) A column of elements in the periodic table.Okay, this is a long, long explanation. Elements Group 18 have a complete outer shell with eight electrons. They are also very reactive because they have seven electrons in the outer shell and will readily accept an electron in order to reach the ion configuration with the ideal number of eight electrons in the outer shell. They all have seven electrons in the outer orbital (two in level s and five in level p). They have only one electron in the outer shell (valence electron) and as a result are highly reactive. All elements in Group 1 for instance are alkali metals. Elements are arranged in these groups according to whose proprieties are similar. Because of the confusion the old IUPAC and the CAS system created, the IUPAC adopted their new system. This method, however, labeled columns 1 and 2 as 'IA' and 'IIA', columns 3 through 7 as 'IIIB' through 'VIB', column 8 through 10 as 'VIII', columns 11 and 12 as 'IB' and 'IIB' and columns 13 through 18 as 'IIIA' through 'VIIIA'. The CAS system, which also used Roman numerals followed by an 'A' or 'B'. 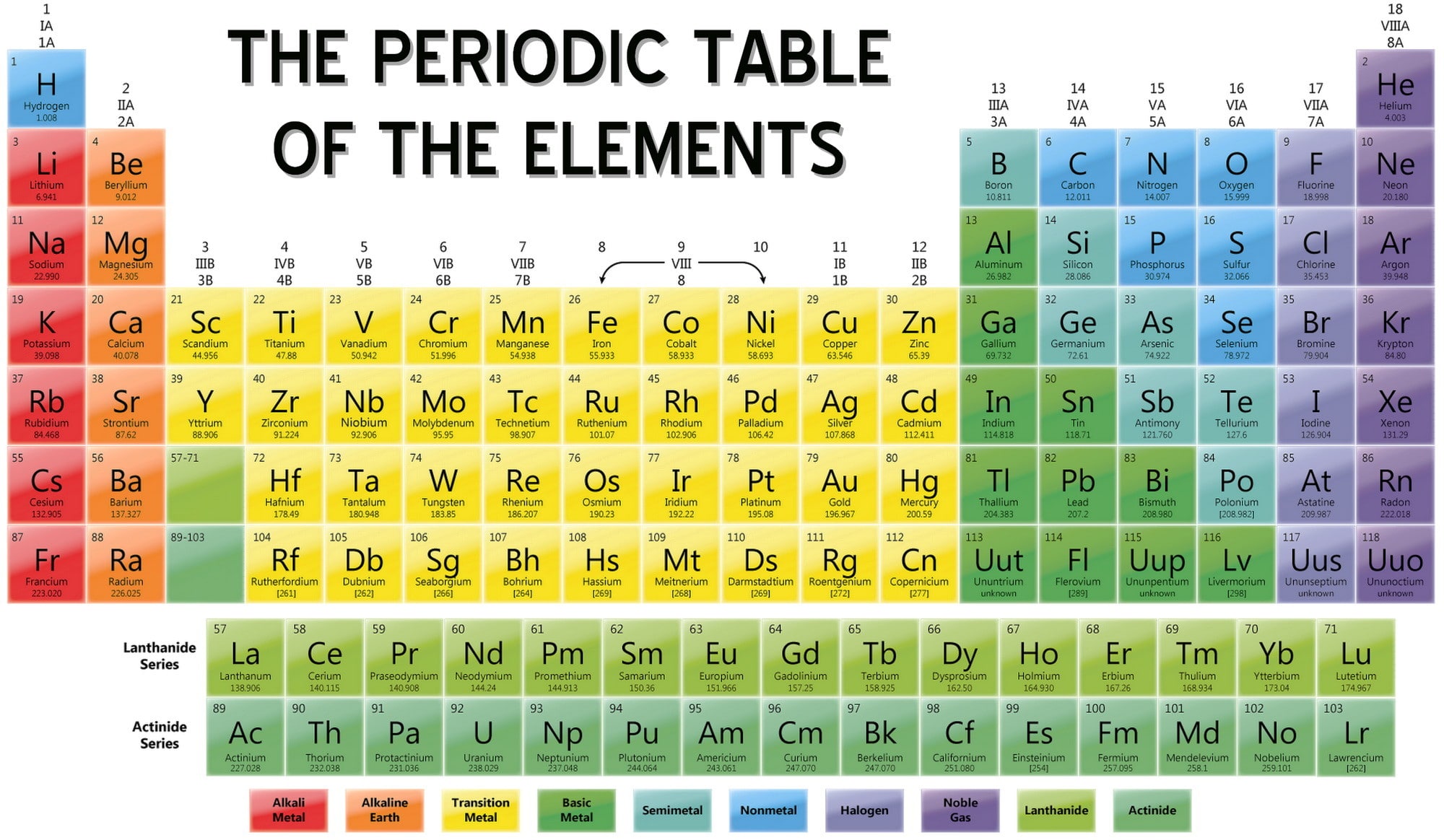 /GettyImages-1154261034-08fa91cb3d8942c093b9e6b66a26f690.jpg)
Columns were numbed such that columns one through seven were numbered 'IA' through 'VIIA', columns 8 through 10 were labeled 'VIIIA', columns 11 through 17 were numbered 'IB' through 'VIIB' and column 18 was numbered 'VIII'. The old IUPAC system, which labeled columns with Roman numerals followed by either the letter 'A' or 'B'. To reduce confusion caused by the other two systems, this is the system that is used in articles on this web site. There are three common numbering systems for these groups: The new IUPAC system, which numbers each column with Arabic numbers from 1 (one) through 18 (eighteen). For more information see Periodic TablesĢ) The vertical columns (major classes or divisions) into which elements are arranged in the periodic table of elements. 1) A vertical column in the periodic table also called a family. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
